
You’ve just taken a course of steroids and are looking for something to normalise your testosterone level. You can’t make up your mind between good old nolvadex, alias tamoxifen, and its cousins raloxifene and toremifene. Greek researchers have published the results of a study in Fertility & Sterility which suggests that tamoxifen is the best choice.
Tamoxifen, raloxifene and toremifene are SERMs: they attach themselves to the receptor for estradiol but don’t start up the processes that usually follow after estradiol has attached itself to its receptor. That’s why doctors use SERMS against tumours whose growth is stimulated by female hormones.
Chemical athletes use SERMs because they raise the testosterone level. One of the ways that the body monitors its own production of sex hormones is by keeping an eye on the concentration of estradiol in the blood. If it gets too high then sex hormone production is reduced – including the production of testosterone. Because the control mechanisms make use of estradiol receptors, SERMs crank up testosterone production. That’s why chemical athletes use substances like tamoxifen after taking a course of steroids.
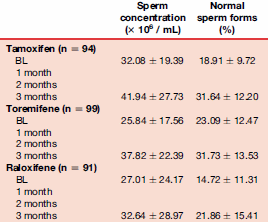
The Greek researchers did a trial with just under three hundred infertile men, whose sperm count was low and most of whom had low levels of testosterone production. The researchers gave the men either 20 mg tamoxifen, 60 mg toremifene or 60 mg raloxifene daily for three months. The table below shows what happened to the men’s LH, FSH and testosterone levels.

Raloxifene has little effect on the testosterone level, so it’s not an interesting candidate for a Post Cycle Therapy supplement. Toremifene is somewhat better, but doesn’t perform as well as tamoxifen, and it loses its maximum effect after two months as well.
To complete the story we’ve added the table below, which shows the effect of the three on sperm cells. Once again, raloxifene performs less well than tamoxifen and toremifene.
The researchers suspect that the two more effective SERMs not only work through the body’s hormonal thermostat, thereby inducing the pituitary gland to make more messenger hormones [which in turn get the testes to produce more testosterone]. They think that tamoxifen and toremifene also have a direct effect on the testosterone producing cells.
The effect of selective estrogen receptor modulator administration on the hypothalamic-pituitary-testicular axis in men with idiopathic oligozoospermia.
Tsourdi E, Kourtis A, Farmakiotis D, Katsikis I, Salmas M, Panidis D.
Source
Second Department of Obstetrics and Gynecology, Division of Endocrinology and Human Reproduction, Aristotle University of Thessaloniki, Thessaloniki, Greece.
Abstract
This study evaluates, compares, and contrasts the effects of three selective estrogen receptor modulators (SERMs), namely, tamoxifen, toremifene, and raloxifene, on the hypothalamic-pituitary-testicular axis in 284 consecutive subfertile men with idiopathic oligozoospermia using three therapeutic protocols: [1] tamoxifen, 20 mg, once daily (n = 94); [2] toremifene, 60 mg, once daily (n = 99); and [3] raloxifene, 60 mg, once daily (n = 91). The antiestrogenic effects of SERMs at the hypothalamic level result in a statistically significant increase of gonadotropin levels, which is more marked for tamoxifen and toremifene compared with raloxifene.
PMID: 18692782 [PubMed – indexed for MEDLINE]
Source: http://www.ncbi.nlm.nih.gov/pubmed/18692782
Nolvadex rejuvenates blood vessels
Chemical athletes who use the anti-oestrogen nolvadex may also be making their blood vessels more healthy, and reducing their risk of a fatal heart attack. We deduce this from a trial done by researchers in Cambridge, England that was published eight years ago in Circulation.
The active ingredient in preparations like nolvadex is tamoxifen [structure shown here]. Nolvadex is a SERM: a compound that is capable of attaching itself to the receptor for the female sex hormone estradiol. What’s more, nolvadex is an anti-oestrogen. The compound forces estradiol off its receptor, thereby preventing estradiol from doing its work. Nolvadex then sits in the estradiol receptor a bit like a key that has broken off in a lock. No other key can be put in the lock, but the door won’t open either. Athletes use nolvadex to counter the side effects of oestrogens, which are produced when testosterone is converted into estradiol. Nolvadex is also used after taking a course of steroids to boost the body’s own testosterone production.
In the British study, 30 men with furred up arteries were given 40 mg of nolvadex every day for 56 days. The researchers wanted to know more about the effect of nolvadex on the heart and the blood vessels. Estradiol has good and bad effects on the heart and arteries. It increases the amount of ‘good cholesterol’ HDL, which is positive. But it also causes blood clots, which is not such good news.
The men’s testosterone levels rose, and their estradiol levels rose too. Apparently the conversion of testosterone into estradiol simply continued.
NCA = 10 male patients with a history of chest pain suggestive of angina pectoris but with normal coronary angiograms. TVD = men with a triple-vessel disease, i.e. clogged up arteries in the heart. This group was given statins and aspirin.
The researchers used a complicated procedure to measure the elasticity of the men’s blood vessels [ED-FMD]. In the graph below you can see that the nolvadex treatment increased the elasticity.
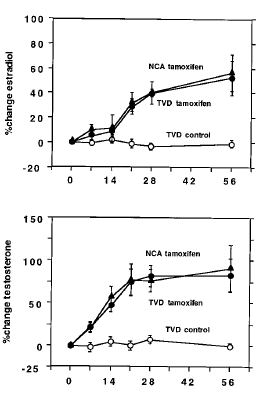
If you’re healthy and under 40, then you probably have an ED-FMD of 7 percent. As you get older your ED-FMD declines to around 5 percent by the age of 51. At your ED-FMD is 3.5 percent. The NCA group had an average age of 51. The average age of the TVD group was 63. So the cardiovascular age of the men decreased.
The figure below shows more cardiovascular parameters.

Most effects are favourable. Lipoprotein (a) is a building block of the ‘bad’ LDL cholesterol. The more of this you have in your blood, the more worried the cardiologists get. The lower the amount of triglycerides and LDL in your blood, the better. They both have an adverse effect on the HDL.
All in all, the researchers were pleasantly surprised by the results of their research. “The data provide strong evidence to support clinical evaluation of SERMs for the treatment of men with CAD”, they write in the last sentence of their article.
Tamoxifen effects on endothelial function and cardiovascular risk factors in men with advanced atherosclerosis
Author(s)
CLARKE Sarah C. (1) ; SCHOFIELD Peter M. (1) ; GRACE Andrew A. (1) ; METCALFE James C. (2) ; KIRSCHENLOHR Heide L. (2) ;
Affiliation(s) du ou des auteurs / Author(s) Affiliation(s)
(1) Department of Cardiology, Papworth Hospital NHS Trust, Papworth Everard, ROYAUME-UNI
(2) Department of Biochemistry, University of Cambridge, Cambridge, ROYAUME-UNI
Abstract
Background-Tamoxifen and its analogues act as selective estrogen receptor modulators (SERMs) in women, with estrogen-like activities on some plasma cardiovascular risk factors (eg, lipoproteins). Effects of SERMs on men with coronary artery disease (CAD) have not been reported. Methods and Results-Thirty-one men with angiographically proven CAD were recruited; 16 were treated with tamoxifen (40 mg/d) for 56 days, and 15 were untreated. All the CAD patients were medicated with aspirin and an HMG-CoA reductase inhibitor for ≥6 weeks before entering the study. Ten men with angina-like symptoms but normal coronary arteries by angiography (NCA group) were also treated with tamoxifen. Blood samples were collected at days -7, 0, 7, 14, 21, 28, and 56 of treatment. Endothelium-dependent flow-mediated dilatation (ED-FMD) of the brachial artery was measured by high-resolution ultrasound at 5 visits. Tamoxifen caused an increase in %ED-FMD maximal at 28 days in the CAD group (2.1±0.3% to 7.5±0.7%; P<0.0001) and the NCA group (3.8±0.4% to 7.9±1.0%; P<0.0001), with no significant change in the untreated group. Tamoxifen also caused decreases in several plasma cardiovascular risk factors, including total cholesterol, triglycerides, lipoprotein(a), and fibrinogen. Except for the triglyceride response, these effects were similar to those reported for postmenopausal women treated with tamoxifen. Conclusions-Tamoxifen substantially increased ED-FMD in men with CAD who were taking conventional medication. Together with the effects on risk factors, the data strongly support clinical evaluation of SERMs for the treatment of men with CAD.



